Kawasaki Disease And Vaccines
Kawasaki disease and vaccines. Prevention of pneumococcal disease among infants and children - use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine - recommendations of the Advisory. Implementation of a standardised Kawasaki disease case definition would increase confidence in the findings and add value to future studies of pre- or post-licensure vaccine safety studies. SIDS Kawasaki Disease and narcolepsy.
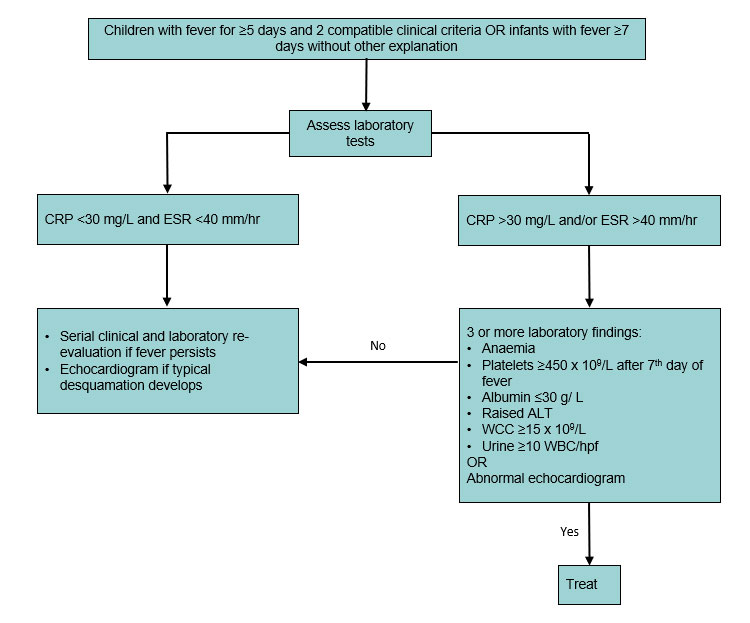
Kawasaki disease KD also known as Kawasaki syndrome is an acute febrile illness of unknown cause that primarily affects children younger than 5 years of age. 2nd episode of Kawasaki Disease NOT refractory disease 4 clinical criteria present. Use of the vaccines was not associated with risk of developing Kawasaki disease low-quality evidence.
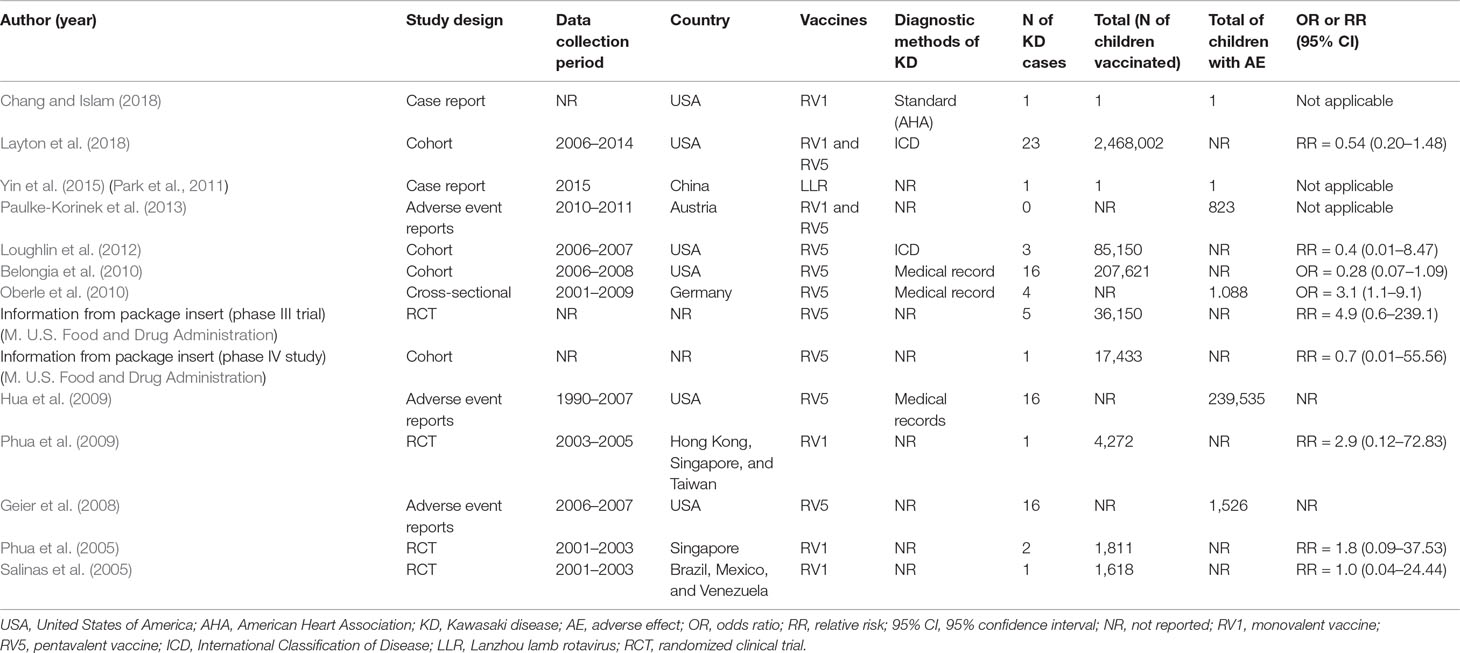
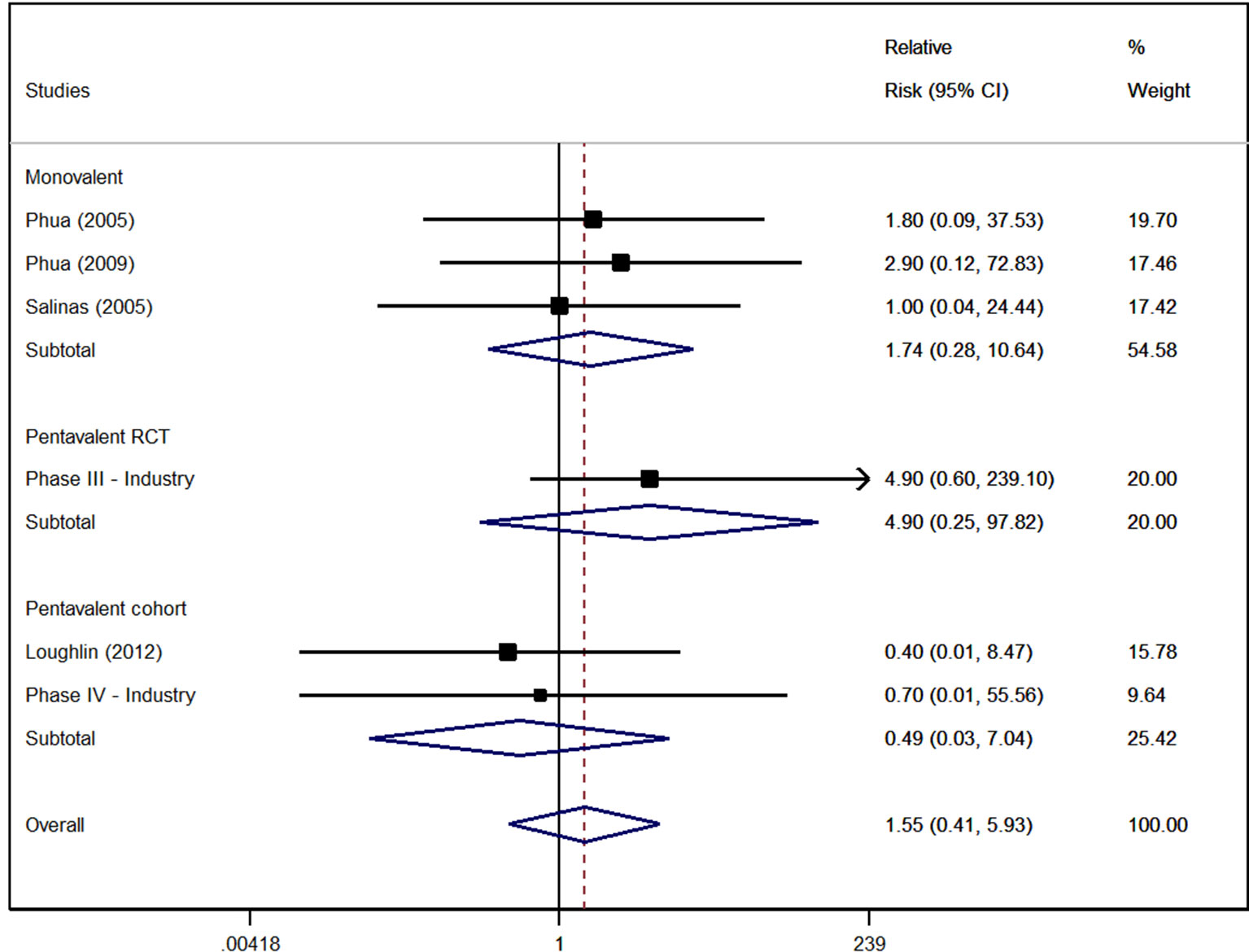
Kawasaki disease first appeared in the 1960s and its frequency in developed nations marched upward right along with the expansion of the immunization schedule. Some childhood shots like the chickenpox vaccine and the vaccine for measles mumps. Since 2006 two rotavirus vaccines have been licensed in Taiwan either as a 2- RV1 or 3-dose RV5 schedule administered at ages 2 4 and 6 months.
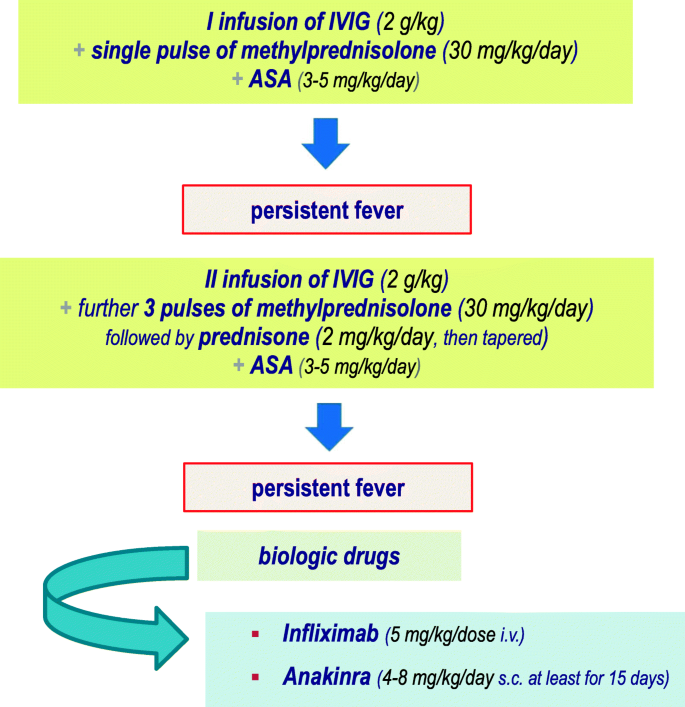
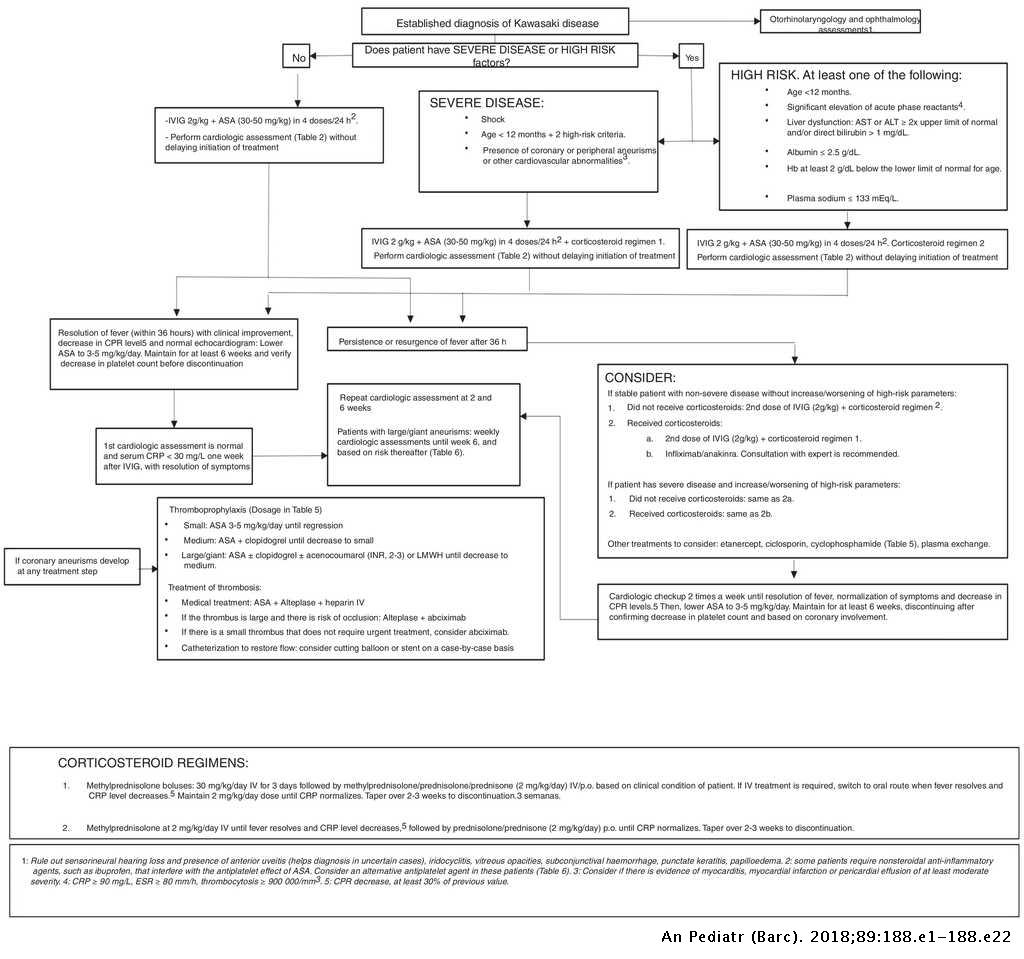
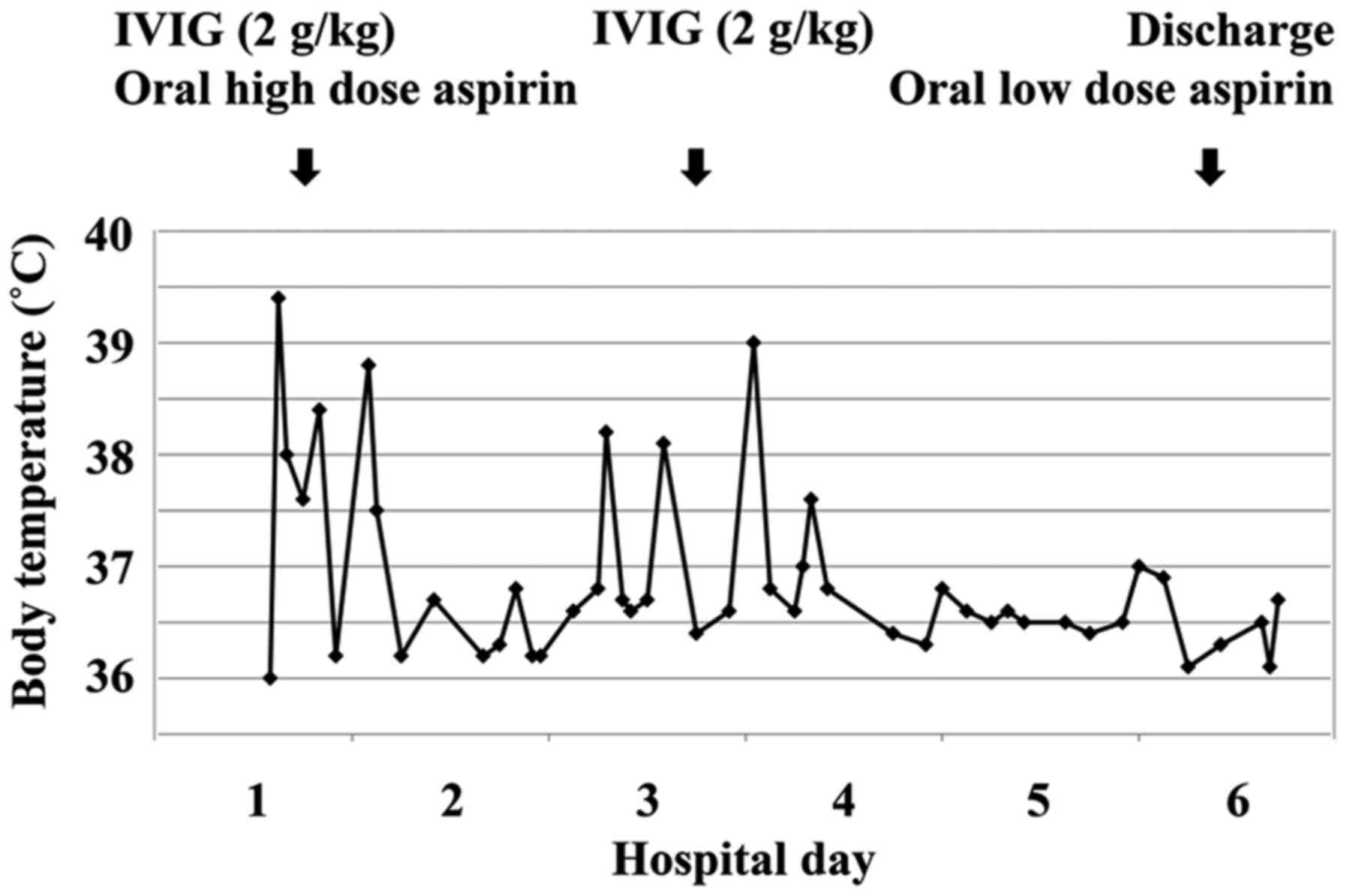
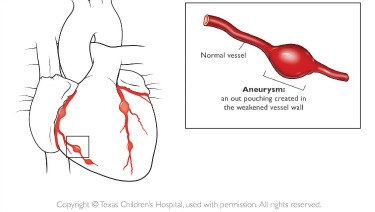
Adjusted incidence rate ratio aIRR 073. In June 2007 the RotaTeq label was revised accordingly. Typically initial treatment of Kawasaki disease consists of high doses of aspirin and immunoglobulin.
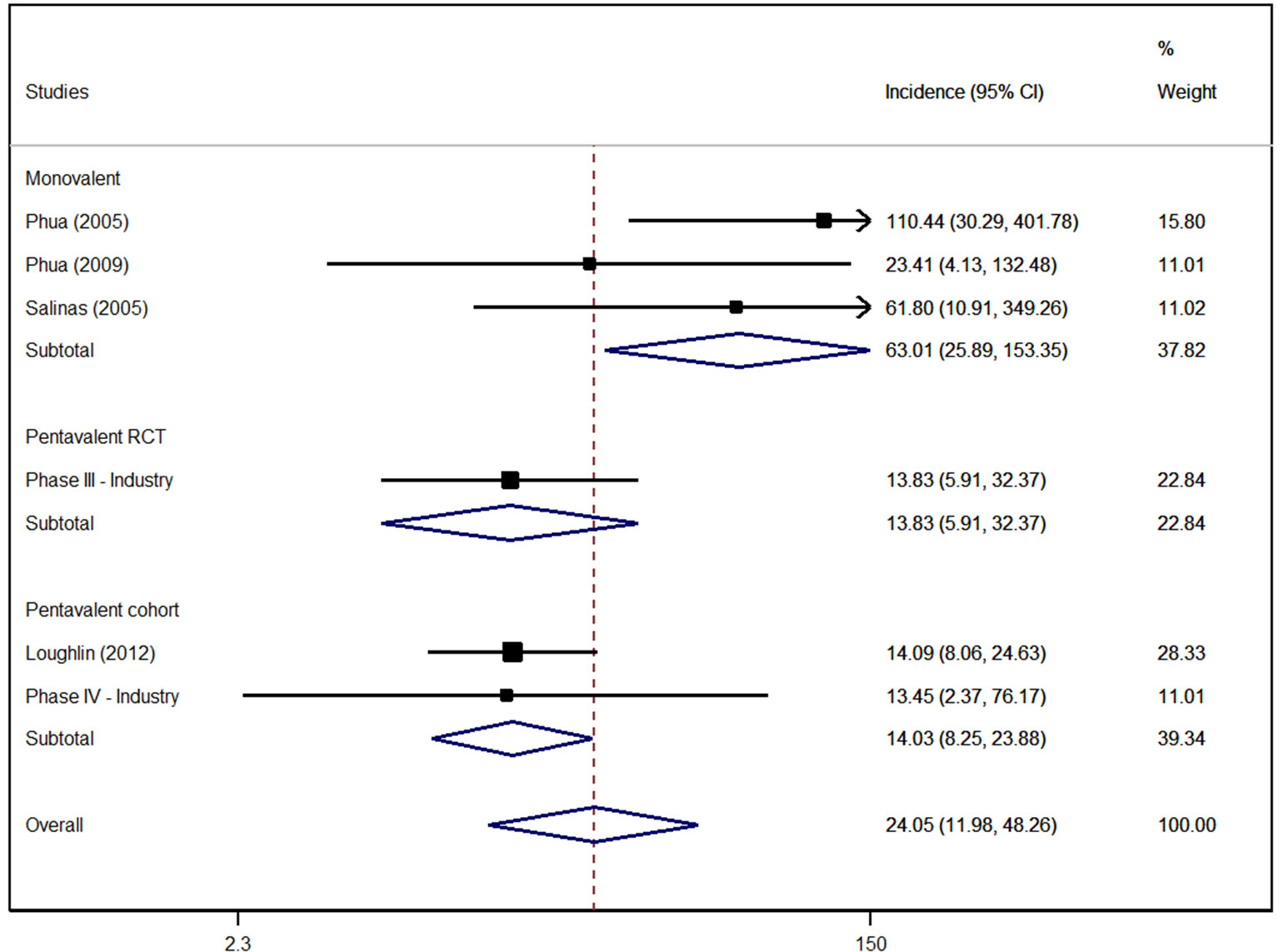
Kawasaki disease KD is a multisystemic vasculitis primarily affecting children vaccine live clinical trial. This study assessed the risk of intussusception and Kawasaki disease KD associated with rotavirus vaccines among infants. Kawasaki disease Unvaccinated 1-21 days 0 0 MIS-C and MIS-A Unvaccinated NA 0 NA Myocarditispericarditis Unvaccinated 1-21 days 2 21 Narcolepsy and cataplexy Unvaccinated NA 2 NA Stroke hemorrhagic Unvaccinated 1-21 days 8 10 Stroke ischemic Unvaccinated 1-21 days 41 388 Transverse myelitis Unvaccinated 1-21 days 0 0.
An emerging Kawasaki-like disease temporally associated with COVID-19 appears to be a distinct syndrome. Your child probably will not get Kawasaki disease again. IVIG is given through a vein over 8 to 12 hours.
Although a temporal relationship between immunisation and Kawasaki disease is suggested evidence for an increased risk or a causal association is lacking. Kawasaki disease KD is a multisystemic vasculitis primarily affecting children vaccine live clinical trial data revealed higher though not statistically significantly KD rates among RotaTeq vaccines than placebo recipients.
Implementation of a standardised Kawasaki disease case definition would increase confidence in the findings and add value to future studies of pre- or post-licensure vaccine safety studies.
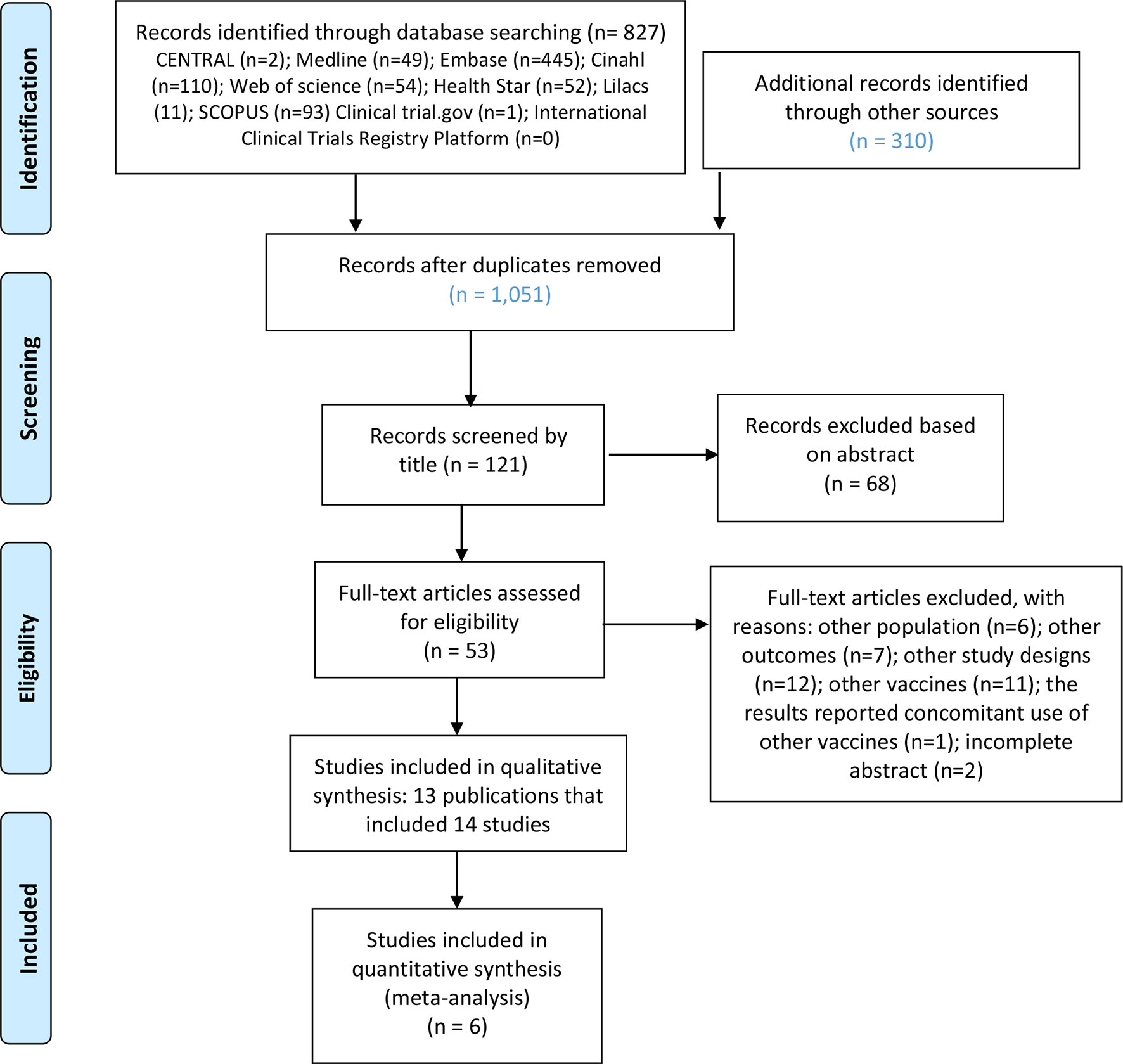
Prevention of pneumococcal disease among infants and children - use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine - recommendations of the Advisory. Kawasaki disease KD also known as Kawasaki syndrome is an acute febrile illness of unknown cause that primarily affects children younger than 5 years of age. Prevention of pneumococcal disease among infants and children - use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine - recommendations of the Advisory. Since 2006 two rotavirus vaccines have been licensed in Taiwan either as a 2- RV1 or 3-dose RV5 schedule administered at ages 2 4 and 6 months. IVIG is given through a vein over 8 to 12 hours. SIDS Kawasaki Disease and narcolepsy. Use of the vaccines was not associated with risk of developing Kawasaki disease low-quality evidence. None of the studies reported the rate of discontinuation of the vaccination schedule. The disease was first described in Japan by Tomisaku Kawasaki in 1967 and the first cases outside of Japan were reported in Hawaii in 1976.
Kawasaki disease KD also known as Kawasaki syndrome is an acute febrile illness of unknown cause that primarily affects children younger than 5 years of age. Prevention of pneumococcal disease among infants and children - use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine - recommendations of the Advisory. Children stay in the hospital for at least 24 hours after completing the IVIG dose to make sure the fever. An emerging Kawasaki-like disease temporally associated with COVID-19 appears to be a distinct syndrome. Usually with treatment fever resolves within 24 hours and full recovery occurs. Your child probably will not get Kawasaki disease again. None of the studies reported the rate of discontinuation of the vaccination schedule.





























Post a Comment for "Kawasaki Disease And Vaccines"